Foreword
Alcohol Flush Reaction (AFR) has been widely studied in the scientific literature, primarily through the lenses of genetics, enzymology, and disease risk. Separately, the biology of glutathione has been extensively examined for its central role in cellular antioxidant defense and detoxification processes.
This white paper brings these established bodies of research together into a single, unified educational framework. Its purpose is not to introduce new clinical claims or propose medical treatments, but to synthesize peer-reviewed findings that help explain why endogenous (naturally produced) glutathione may be an important consideration in the context of Alcohol Flush Reaction and acetaldehyde-associated oxidative stress.
While the underlying biochemical mechanisms discussed in this paper have been studied independently for decades, they are rarely presented together within an AFR-focuses analysis. This document is intended to bridge that gap by translating existing science into a coherent, accessible overview for educational and informational purposes.
This white paper is not medical advice and does not diagnose, treat, cure, or prevent any disease. Readers are encouraged to consult qualified healthcare professionals regarding individual health decisions.
Disclosure: While Alcohol Flush Reaction(AFR) is used throughout this paper as an established reference term, the Addendum provides updated clarification regarding the underlying genetic condition and terminology.
Alcohol Flush Reaction (AFR): Genetic and Metabolic Mechanisms of Impaired Acetaldehyde Clearance
White Paper by Yosh Nakano
Independent Researcher
January 25, 2026
Category Definition
Alcohol Flush Reaction (AFR) is a metabolic condition characterized by impaired acetaldehyde clearance resulting from reduced aldehyde dehydrogenase 2 (ALDH2) activity. This impairment leads to acetaldehyde accumulation following alcohol exposure and may also contribute to symptoms associated from endogenous acetaldehyde production independent of ethanol intake.
Origins
The Short Answer
AFR originated as an evolutionary adaptation caused by a genetic mutation (ALDH2 deficiency) that spread though East Asia because it likely conferred survival advantages during ancient disease and environmental stress periods.
The Long Answer (what science suggests)
- The mutation appeared thousands of years ago
- Genetic evidence dates the ALDH2 mutation to roughly 2,000-3,000 years ago
- It emerged in East Asia, particularly:
- Southern China
- The Chang Jiang basin
- From there, it spread to populations in:
- China
- Korea
- Japan
- Vietnam
- Parts of Southeast Asia
2. The “disease pressure” hypothesis (what you read about)
Researchers believe the mutation spread during periods when infectious disease, contaminated water, and food-borne pathogens were widespread due to:
- Dense agricultural settlements
- Rice cultivation and irrigation
- Early fermentation of grains
- Limited sanitation
Why this matters:
- Alcohol consumption increased acetaldehyde exposure
- Acetaldehyde is toxic to microbes
- People with impaired acetaldehyde clearance experienced immediate negative reactions to alcohol
Those reactions discouraged heavy drinking, which may have:
- Reduced risky behaviors
- Lowered exposure to contaminated alcohol/water
- Preserved immune and liver function during epidemics
3. Selective advantage (counterintuitive but real)
Although AFR feels like a disadvantage today, historically it may have:
- Reduced alcoholism
- Improved survival during famines or epidemics
- Preserved reproductive success
This explains why the mutation:
- Spread rapidly
- Persisted strongly in specific regions
- Did not disappear over generations
4. Why it stayed geographically concentrated
- The mutation arose after major human migrations out of Africa
- It spread regionally, not globally
- Populations outside East Asia had no selective pressure to retain it
5. Modern mismatch: ancient genetics, modern alcohol
Today, the environment has changed:
- Alcohol is ubiquitous
- Social drinking is normalized
- Acetaldehyde exposure is higher and more frequent
But the genetic limitation remains, which is why AFR is now viewed as:
- A genetic metabolic condition
- Not a cultural issue
- Not a racial label
- Not a disease you “catch”
Why Alcohol Flush Reaction (AFR) can occur without alcohol
Core Principle
AFR is not caused by alcohol itself. It is caused by insufficient clearance of acetaldehyde, a reactive aldehyde that is produced endogenously through multiple biochemical pathways.
Alcohol is merely the most visible trigger, not the only source.
1. Acetaldehyde is produced endogenously (baseline physiology)
Acetaldehyde is continuously generated in the human body from:
- Normal cellular metabolism
- Lipid peroxidation
- Oxidative stress
- Inflammatory processes
Even in abstinent individuals, measurable acetaldehyde levels are present in blood and tissues.
AFR represents a failure to detoxify both exogenous and endogenous acetaldehyde.
2. Gut microbiome—derived acetaldehyde (major non-alcohol source)
Certain gut and oral microbes possess alcohol dehydrogenase (ADH) activity and can convert:
- Dietary sugars
- Fermented foods
- Endogenous ethanol (produced by gut fermentation) into acetaldehyde.
This process occurs:
- Without drinking alcohol
- Especially in individuals with:
- Dysbiosis
- High-carbohydrate diets
- Small intestinal bacterial overgrowth (SIBO)
In individuals with ALDH2 deficiency, this acetaldehyde is poorly cleared, triggering AFR-like symptoms.
3. Oxidative stress increases aldehyde burden
Reactive oxygen species (ROS) generate aldehydes such as:
- Acetaldehyde
- Malondialdehyde
- 4-hydroxynonenal (4-HNE)
These aldehydes:
- Compete for ALDH2 detoxification capacity
- Exacerbate mitochondrial stress
- Amplify vasodilation and flushing responses
Thus, stress, illness, smoking, air pollution, or inflammation can provoke AFR symptoms independent of alcohol intake.
4. ALDH2 deficiency creates a systemic aldehyde bottleneck
In individuals with the ALDH2*2 polymorphism:
- ALDH2 enzymatic activity is reduced by ~70-90%
- Detoxification capacity is easily overwhelmed
- Even small aldehyde increases trigger:
- Flushing
- Tachycardia
- Headache
- Nausea
This reframes AFR as a chronic metabolic limitation, not an episodic alcohol reaction.
5. Endogenous glutathione dependence increases in ALDH2 deficiency
When ALDH2 capacity is limited, the body increasingly relies on:
- Glutathione-dependent detoxification
- Aldehyde conjugation
- Redox buffering in mitochondria
This explains why AFR is intricately linked to:
- Mitochondrial oxidative stress
- Depletion of endogenous glutathione
- Heightened sensitivity to non-alcohol aldehyde sources
The Acetaldehyde Mechanism (Core AFR Pathophysiology)
Alcohol Flush Reaction (AFR) originates from impaired acetaldehyde metabolism resulting from reduced aldehyde dehydrogenase 2 (ALDH2) activity, leading to acetaldehyde accumulation and engagement of secondary detoxification pathways.
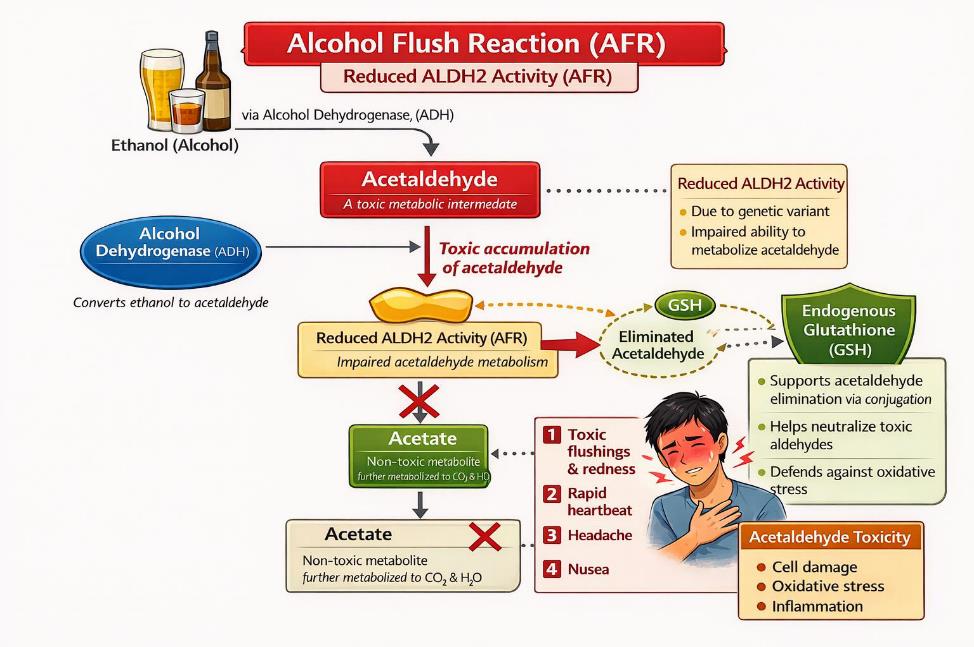
Figure 1. Alcohol Flush Reaction (AFR): Impaired Acetaldehyde Metabolism and Compensatory Glutathione Conjugation
Step 1: Ethanol is rapidly converted to acetaldehyde
- Alcohol (ethanol) is metabolized primarily by alcohol dehydrogenase (ADH).
- This step occurs efficiently in most people, including those with AFR.
Result: Acetaldehyde is produced quickly.
Step 2: Impaired conversion of acetaldehyde to acetate
- In individuals with AFR, aldehyde dehydrogenase 2 (ALDH2) activity is reduces due to a genetic variant.
- This limits the conversion of acetaldehyde into acetate, a relatively non-toxic metabolite.
Result: Acetaldehyde accumulated
Step 3: Acetaldehyde accumulation drives toxicity
Acetaldehyde is:
- Highly reactive
- Capable of forming DNA and protein adducts
- A potent inducer of oxidative stress
- A known toxic and carcinogenic metabolite
Result: Cellular stress, vascular effects, and characteristic AFR symptoms (flushing, tachycardia, nausea, headache)
Step 4: Compensatory detoxification pathways are engaged
When ALDH2-mediated clearance is insufficient.
- The body relies more heavily on secondary detoxification mechanisms
- Endogenous glutathione (GSH) contributes by:
- Conjugating acetaldehyde
- Supporting antioxidant defense
- Limiting oxidative damage
Important: This pathway is slower and less efficient than normal ALDH2 function.
Step 5: Prolonged exposure amplifies downstream effects
If acetaldehyde clearance remains delayed:
- AFR symptoms persist
- Oxidative stress increases
- Cellular and tissue-level effects become more pronounced
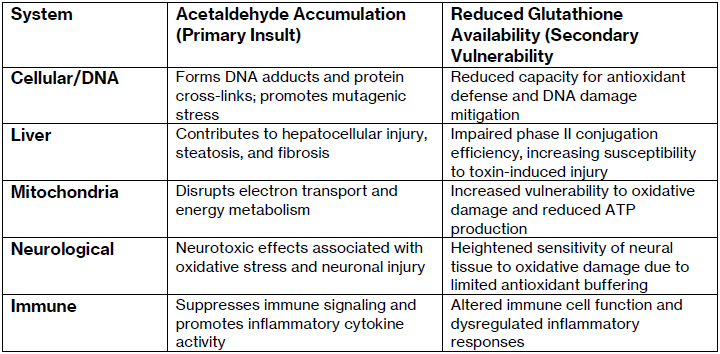
The Harmful Effects of Acetaldehyde
Acetaldehyde is a highly reactive and toxic metabolic intermediate. When clearance is impaired, prolonged exposure can disrupt multiple cellular systems and contribute to downstream health risks.
Cellular & DNA Damage
- Formation of DNA adducts & mutagenic lesions
- Protein cross-linking that impairs normal cellular function
- Increased oxidative stress and cellular injury
Carcinogenic Effects
- Elevated risk of esophageal & head and neck cancers
- Genomic instability linked to acetaldehyde-induced DNA damage
Liver Toxicity
- Contribution to fatty liver disease, hepatitis, and cirrhosis
- Mitochondrial disfunction affecting hepatic energy metabolism
Cardiovascular & Cognitive
- Endothelial damage and impaired vascular function
- Associations with hypertension, atherosclerosis, and cardiomyopathy
Immune & Inflammation
- Immune suppression
- Cytokine-mediated inflammatory responses
Gastrointestinal Effects
- Gastritis mucosal irritation and inflammation
- Increased intestinal permeability and microbiome disruption
Physiological Consequences of Reduced Glutathione Availability
- Increased oxidative stress and reactive oxygen species accumulation
- Reduced efficiency of cellular detoxification pathways
- Impaired handling of electrophilic toxins and environment contaminants
- Increased susceptibility to oxidative DNA damage
- Altered cellular membrane integrity
- Disruption of normal cellular energy metabolism
- Increased cellular vulnerability to injury under stress conditions
Important AFR Facts
- Alcohol Flush Reaction (AFR) is associated with a genetic variant affecting ALDH2, resulting in reduced acetaldehyde metabolism.
- A single copy of the variant can substantially reduce enzyme activity and trigger AFR symptoms.
- AFR is not an allergy
- AFR is not caused by alcohol intolerance originating in the gastrointestinal tract.
- AFR is a cellular detoxification limitation, specifically impaired acetaldehyde clearance.
Why AFR matters beyond flushing?
Modern scientific understanding emphasizes the importance of the body’s endogenous detoxification systems particularly those involved in acetaldehyde clearance downstream of alcohol metabolism. Endogenous glutathione is a secondary/compensatory pathway, especially when ALDH2 is impaired.
In individuals with reduced ALDH2 activity, acetaldehyde elimination relies more heavily on slower, secondary detoxification mechanisms. Endogenous glutathione (GSH) contributes to acetaldehyde conjugation and supports cellular defense against oxidative stress, however, these pathways are less efficient than normal ALDH2-mediated metabolism, allowing acetaldehyde to accumulate and prolong symptoms.
The Mirror Effects: Acetaldehyde vs. GSH Deficiency
Why They Feel the Same
When acetaldehyde levels rise—such as after alcohol consumption or through endogenous production associated with gut dysbiosis—it is cleared in part through glutathione-dependent conjugation reactions. Increased acetaldehyde burden can accelerate glutathione utilization, temporarily reducing its availability for other antioxidant and detoxification functions.
As a result, symptoms commonly attributed to elevated acetaldehyde exposure—such as headache, cognitive fog, and systemic discomfort—may overlap with those associated with reduced glutathione availability and increased oxidative stress. This shared biochemical burden helps explain why acetaldehyde accumulation and glutathione depletion can produce similar physiological effects.
Consequently, the symptoms of “Acetaldehyde Overload” (hangovers, brain fog, liver pain) are actually the symptoms of “Acute Glutathione Bankruptcy.”
Summary
Alcohol Flush Reaction (AFR) is associated with reduced activity of aldehyde dehydrogenase 2 (ALDH2), resulting in impaired acetaldehyde metabolism. In affected individuals, acetaldehyde accumulation can occur not only following alcohol consumption
but also through endogenous sources related to normal metabolism, gut microbial activity, oxidative stress, and lipid peroxidation. This persistent acetaldehyde burden contributes to characteristic AFR symptoms independent of ethanol exposure.
When ALDH2-mediated clearance is limited, secondary detoxification and redox-buffering systems play a greater compensatory role. Endogenous glutathione-dependent pathways contribute to the conjugation and clearance of reactive aldehydes and help mitigate oxidative stress. However, these pathways are less efficient than normal ALDH2 activity and may become strained under conditions of increased acetaldehyde exposure.
These findings underscore the role of acetaldehyde accumulation and downstream detoxification capacity in individuals with Alcohol Flush Reaction. Continued investigation into endogenous antioxidant and aldehyde-handling systems may help clarify variability in AFR-associated physiology and symptom.
Alcohol Flush Reaction Sources
I. Core sources on the origin and spread of AFR (ALDH2 deficiency)
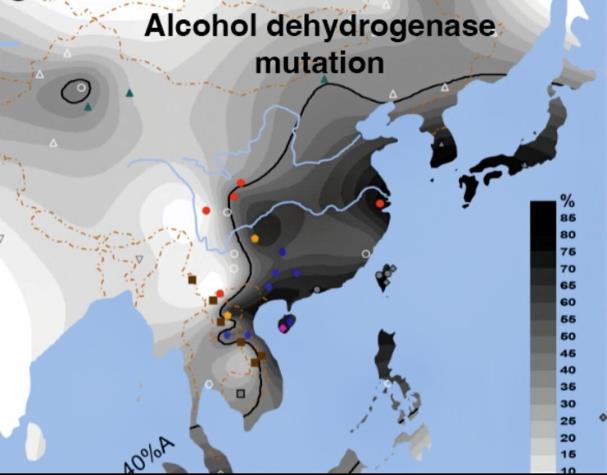
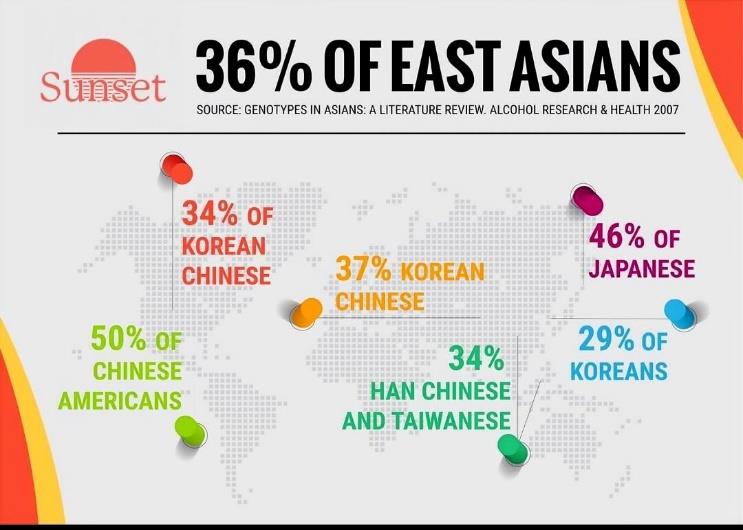
1. Li et al., 2009—PloS ONE
Title: Worldwide Human Populations and ALDH2 Gene Polymorphism
Why this matters:
- Definitively maps ALDH2*2 geographic distribution
- Shows high prevalence in East Asia and near absence elsewhere
Citation:
Li, H. et al. (2009). PloS ONE, 4(4): e5312.
Doi: 10.1371/journal.pone.0005312)
2. Brooks et al., 2009—Pharmacology & Therapeutics
Title: The Alcohol Flushing Response: An Unrecognized Risk Factor for Esophageal Cancer
Why this matters:
- Comprehensive review of ALDH2 deficiency
- Explains acetaldehyde toxicity
- Provides strong mechanistic framing without racialized language
Citation:
Brooks, P.J. et al. (2009). Pharmacol Ther., 123(2), 238-247
Doi:10.1016/j.pharmtherea.2009.04.002
3. Peng & Yin, 2009—Human Genomics
Title: The ALDH2*2 Allele: Global Distribution and Evolutionary Origin*
Why this matters:
- Directly addresses evolutionary hypotheses
- Proposes selective advantage models
- Often cited when discussing ancient disease/environmental pressures
Citation: Peng, G.S., & Yin, S.J. (2009). Human Genomics, 3(3), 281-288
4. Chih-Lang Lin, et Al, Sept 2021 Biomolecules
Title: The Aldehyde Dehydrogenase ALDH2*2 Allele, Associated with Alcohol Drinking Behavior, Dates Back to Prehistoric Times
Citation: Chih-Lang Lin et al. (2021). Biomolecules 2021, 11(9), 1376 Doi.org/10.3390/biom11091376
II. Sources connecting AFR to ancient disease & environmental pressure
5. Oota et al., 2004—American Journal of Human Genetics
Title: The Evolution of ALDH2 Polymorphism and Population History in East Asia
Why this matters:
- Genetic dating places mutation at ~2,000-3,000 years ago
- Correlates spread with rice agriculture expansion
- Strong evolutionary anthropology foundation
Citation: Oota, H. et al. (2004). Am J Hum Genet., 75(3), 441-450.doi:10. 1086/423901
III. Sources on mechanism (ALDH2, acetaldehyde, oxidative stress)
6. Edenberg, 2007—Nature Reviews Genetics
Title: The Genetics of Alcohol Metabolism
Why this matters:
- Authoritative overview of ethanol->acetaldehyde->acetate
- Often cited as a foundational reference
Citation:
Edenberg, H. J. (2007). Nat Rev Genet., 8, 845-858
doi.10.1038/nrg2162
7. Setshedi et al., 2010—Alcohol Research & Health
Title: Acetaldehyde, Oxidative Stress, and Mitochondrial Injury Why this matters:
- Connects acetaldehyde accumulation to mitochondrial dysfunction
- Useful bridge to endogenous detoxification systems
Citation: Oxidative Medicine & Cellular Longevity 2010 May-Jun;3(3):178-85. doi: 10.4161/oxim.3.3.12288
Non-alcohol Acetaldehyde Sources
Endogenous (human metabolism/oxidative stress) acetaldehyde
1. Lipid peroxidation can generate free acetaldehyde in cells
Pospisil P, et al. Formation of free acetaldehydes derived from lipid peroxidation in U937 monocyte-like cells. Biochim Biophys Acta Gen Subj. 2024, 1868:130527. doi: 10.1016/j.bbagen.wo23.130527 Formation of free acetaldehydes derived from lipid peroxidation in U937 monocyte-like cells – PubMed
2. Threonine catabolism produces acetaldehyde endogenously (useful as a “baseline endogenous source” statement) Garcia CCM, et al. [12C2]-Acetaldehyde promotes unequivocal formation of DNA adducts…J Am Chem Soc. 2011. (States endogenous acetaldehyde arises during Threonine catabolism) doi: 10.1021/ja2004686 [13C2]- Acetaldehyde Promotes Unequivocal Formation of 1,N2-Propano-2′-deoxyguanosine in Human Cells | Journal of the American Chemical Society
3. Direct experimental evidence of acetaldehyde from a physiological source (threonine) in non-alcoholic liver injury context Ma XL, Baraona E, Hernandez-Munoz R, Lieber CS. High levels of acetaldehyde in nonalcoholic liver injury after threonine or ethanol administration. Hepatology. 1989;10(6):933-940. doi: 10.1002/hep.1840100607 High levels of acetaldehyde in nonalcoholic liver injury after threonine or ethanol administration – PubMed
4. When aldehyde detox is impaired, endogenous aldehydes accumulate (genotoxic burden) Antonowicz S, et al. Endogenous aldehyde accumulation…Nat Commun. 2021 (Not acetaldehyde-only; strong for the broader point that aldehyde detox defects elevate endogenous aldehyde/genotoxin load.) Endogenous aldehyde accumulation generates genotoxicity and exhaled biomarkers in esophageal adenocarcinoma | Nature Communications
Microbial acetaldehyde without drinking alcohol
5. Oral microbes can produce acetaldehyde from glucose fermentation (no beverage alcohol required) Nieminen MT, Uittamo J, Salaspuro M, Rautemaa R. Acetaldehyde production from ethanol and glucose by non-Candida albicans yeasts in vitro. Oral Oncol. 2009;45(12):e245-e248. doi: 10.1016/j.oraloncology.2009.08.002 Acetaldehyde production from ethanol and glucose by non-Candidaalbicans yeasts in vitro – ScienceDirect
6. Local (oral cavity) acetaldehyde exposure can occur independently of hepatic metabolism (useful when arguing “site-specific acetaldehyde can be generated locally”) Stornetta A, Guidolin V, Balbo S. Acetaldehyde Exposure in the Oral Cavity. Cancers (Basel). 2018;10(1):20. Alcohol-Derived Acetaldehyde Exposure in the Oral Cavity – PMC
Harmful Effects of Acetaldehyde
1. Cellular & DNA Damage
- DNA Adducts & Mutations: Acetaldehyde is classified as a Group 1 Carcinogen by the IAC. It reacts with DNA to form bulky adducts (like N2-ethyl-2’-deoxyguqnosine), which lead to permanent mutations and genomic instability.
- Citation: Brooks, P.J., & Theruvathu, J. A. (2005). “DNA adducts from acetaldehyde: implications for alcohol-related carcinogenesis.”
Mutation Research:
- Protein Cross-linking: It acts as an electrophile, binding to proteins to form “acetaldehyde adducts” that impair enzyme function and trigger autoimmune responses.
- Citation: Tuma, D. J., & Casey, C.A. (2003). “Dangerous Liaisons: Fatty Acids and Alcohol-Induced Tissue Damage.” Alcohol Research & Health.
- Citation: Alcohol-Induced Oxidative Stress and the Role of Antioxidants in Alcohol Use Disorder: A Systematic Review – PMC
- Citation: Yokoyama, A., et al. (2010). “Genetic polymorphisms of alcohol-metabolizing enzymes and upper aerodigestive tract cancers.”
2. Cancer Science
- Citation: Song, K., et al. (2022). Acetaldehyde induces Cytotoxicity via Triggering Mitochondrial Dysfunction. Molecules.
- Citation: Alcohol Consumption, ALDH2 Polymorphism as Risk Factors for Upper Aerodigestive Tract Cancer Progression and Prognosis
3. Liver Toxicity
- Citation: Exploring Cirrhosis: Insights into Advances in Therapeutic Strategies
- Citation: Setshedi, M., et al. (2010). “Acetaldehyde adducts in alcoholic liver disease.” Oxidative Medicine and Cellular Longevity.
4. Cardiovascular & Cognitive
- Hypertension & Endothelial Damage: It disrupts the vascular lining (endothelium), leading to stiffness and high blood pressure.
- Citation: Zhang, Y., et al. (2010). “Acetaldehyde and its cardiovascular toxicity.” Cardiovascular Toxicology.
- Cytokine-driven inflammation: Acetaldehyde activates the NF-kB signaling pathway, which triggers the release of pro-inflammatory cytokines like IL-1B and TNF-a.
- Citation: Wang, H.J., et al. (2005). “Acetaldehyde-induced interleukin-1beta and tumor necrosis factor-alpha production.” PubMed.
- Immune Suppression: While it triggers inflammation, it simultaneously impairs the ability of white blood cells to clear pathogens, increasing infection risk.
- Citation: Focus on: Alcohol and the immune system.
5. Gastrointestinal Effects
Disclosure/Conflict of Interest
The author is the owner and president of MagicalGSH LLC, a Nevada company based company operating in the nutritional supplement sector.
This white paper is an independent, non-commercial synthesis of publicly available scientific literature. No products, therapies, or commercial interventions are discussed or promoted. The analysis and interpretations presented are intended solely for educational and scientific discussion.
No part of this document may be reproduced, distributed, or transmitted in any form or by any means without prior written permission of the author, except for brief quotations used for scholarly or educational purposes with appropriated attribution.
© 2026 Yosh Nakano. All Rights Reserved.